The 1974 Aluminum Penny: The Coin That Was Never Meant to Exist
Copper prices went really high in the 1970s.
This made it costly to make pennies; the cost of making one penny was actually more than the penny was worth.
Aluminum was a choice because it was cheap and easy to get.
So, they did some tests with aluminum in 1973 to see if it would work for making pennies.
The tests were to see if aluminum could be used to make pennies of copper.
Engineers aimed to create an alloy possessing sufficient durability for long-term 1974 aluminum penny circulation.
Characteristic | Standard Copper | Experimental Aluminum |
Composition | 95% copper, 5% zinc | 99% aluminum with additives |
Coin Weight | 3.11 g | 0.91 g |
Production Cost | $0.009 | $0.0003 |
Wear Resistance | High | Medium |
Description and Physical Properties
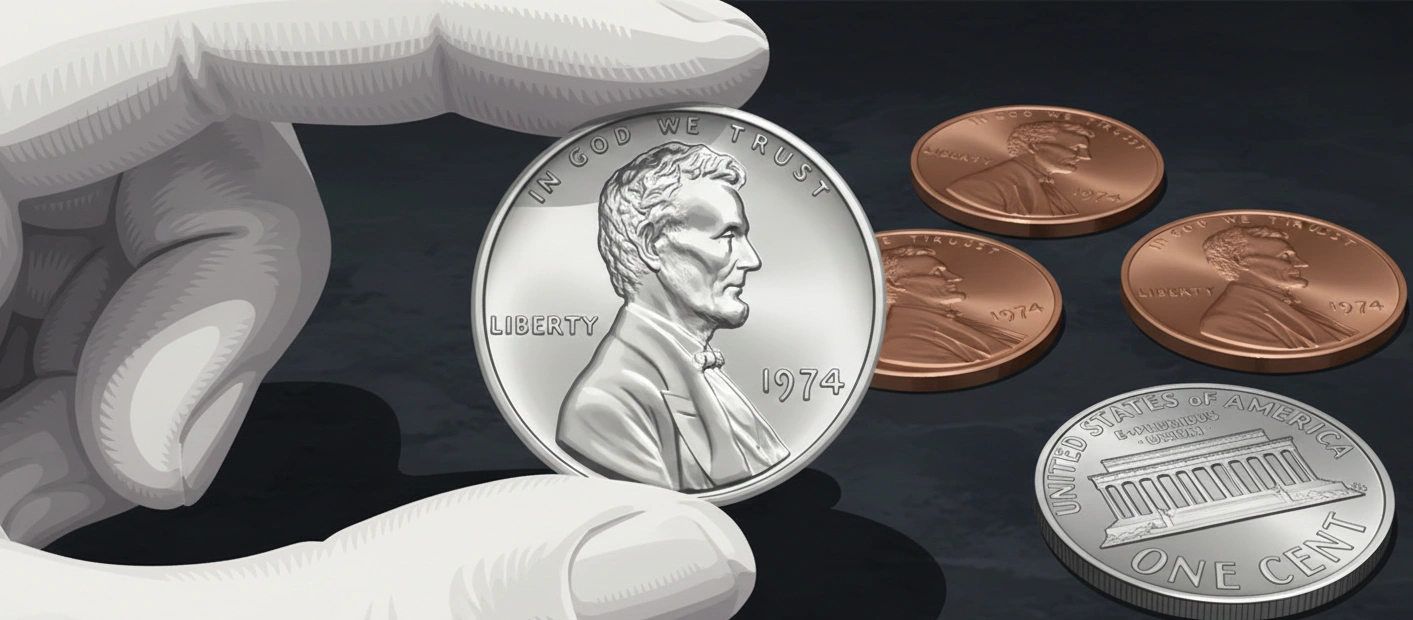
This experimental coin retained the design of the standard Lincoln cent.
Using aluminum resulted in a 71% weight reduction compared to the copper counterpart.
Low weight created difficulties when operating vending machines calibrated for specific density and mass parameters.
Coining equipment required pressure adjustments owing to the softness of aluminum.
These coins possessed a bright silver-white luster, distinguishing them from traditional bronze cents.
The metal surface demonstrated a tendency toward rapid oxidation when contacting aggressive environments.
Parameter | Value |
Diameter | 19.05 mm |
Thickness | 1.55 mm |
Mass | 0.911 g |
Edge Type | Plain |
Production Figures and Sample Distribution
The Philadelphia Mint produced a batch of 1,579,324 specimens in late 1973.
Dating on all struck coins corresponded to the year 1974.
Mint officials distributed samples to members of the U.S. Congress for review and assessment of the reform's feasibility.
The transfer procedure occurred without a strict protocol for tracking and registering each serial number.
Most legislators failed to return the specimens received after the conclusion of the hearings.
Department staff demanded the return of all samples following the official cancellation of the program in 1974.
Distribution Statistics
Total Mintage: 1,579,324 units
Destroyed under supervision: ~1,579,300 units
Distributed to officials: ~25 units
Officially confirmed existence: 2 units
Status of remaining specimens: Unknown
Program Cancellation and Legal Consequences
The U.S. radiological community opposed the introduction of aluminum coins.
Doctors argued their protest based on the impossibility of detecting such objects on X-rays if accidentally swallowed by children.
Aluminum possesses low radiopacity compared to copper or zinc.
The Mint recognized the technical risks and complexities of recalibrating vending machines as being too high.
Official rejection of aluminum led to the destruction of nearly the entire mintage in smelting furnaces.
Legal status regarding the surviving coins remains a subject of dispute between the government and private owners.
All 1974 aluminum cents are considered U.S. government property, not subject to legal private ownership.
Description of Known Specimens and Their Value
One confirmed specimen resides in the Smithsonian Institution as part of the National Numismatic Collection.
A second known coin is linked to the name of Albert Treddle, a former Capitol Police officer.
Treddle received the coin from a congressman dropping it in the Capitol building in 1974.
Treddle's heirs discovered the coin in 2013, initiating the certification process.
The PCGS laboratory confirmed the specimen's authenticity, assigning it a grade of Mint State 66.
The U.S. government asserted claims of ownership over this coin, forcing the family to return it to the state in 2014.
Potential Auction Value Estimates
The market price for a 1974 aluminum cent remains hypothetical due to the ban on trading.
Experts base forecasts on the value of similar rarities, such as the 1943 copper cent.
Price growth dynamics for unique U.S. coins average between 7% and 12% annually.
Sales Scenario | Estimated Value | Potential Annual Growth |
Private transaction | $200,000 – $400,000 | 5% |
Official public auction | $2,000,000 – $3,500,000 | 10% |
Sale as part of a collection | $1,500,000+ | 8% |
A drop in value is excluded because of the physical lack of supply on the market.
Inflationary processes increase the nominal price of such objects by 3% in the long term.
Metallurgical Analysis and Preservation Issues
The 1974 aluminum alloy exhibits high porosity during macro photography.
The micro-defects are transferred to the coin surface with high clarity because of the material's plasticity.
Lacking the patina characteristic of copper makes aluminum coins vulnerable to mechanical damage.
Aluminum's hardness on the Mohs scale is approximately 2.75.
Copper alloys possess a hardness of about 3.0, providing better protection against scratches.
Chemical analysis confirms the absence of toxic impurities in the experimental composition.

Denver Aluminum Cent (1974-D)
The existence of an aluminum cent with the "D" mint mark was long considered a numismatic myth.
A Denver Mint employee kept one specimen after discovering it in production waste.
Federal agents seized the coin following an attempt at its appraisal in 2016.
Denver Mint documentation contains no records of mass striking aluminum planchets.
Likely, the Denver batch was limited to a few test strikes for equipment verification.
This specific specimen holds the status of physical evidence within the Secret Service archives.
Impact on Subsequent Monetary Policy
Failure of the aluminum experiment forced the pursuit of other ways to reduce the cent.
The Mint transitioned to using zinc planchets with a thin copper coating in 1982.
A zinc cent weighs 2.5 grams, occupying a middle ground between copper and aluminum.
Switching to zinc allowed for a 40% reduction in costs without losing coin functionality.
The 1974 experience demonstrated the importance of interacting with the medical community when implementing new materials.
Using aluminum in U.S. coinage was no longer considered a viable option.
Technological Aspects of Disposal
The disposal process for the aluminum mintage was carried out in induction furnaces.
Aluminum's high melting point enabled rapid processing of the mass into ingots.
Recycled aluminum was returned to the industrial cycle for manufacturing non-monetary items.
U.S. Secret Service personnel supervised the destruction.
Lacking destruction certificates for each specific specimen creates a legal loophole for searching for lost coins.
Inspectors recorded the total mass of destroyed metal, matching the design calculations.



